In September 2020, Dr. Anthony Fauci declared in an interview that the U.S. Food and Drug Administration's Data and Safety Monitoring Board (DSMB) "is an independent body." Clarifying further, he explained that the board—which offers expert advice to the FDA on a range of issues, including the approval of the Pfizer jab—is "beholden to no one, not to the president, not to the vaccine companies, not to the FDA. Not to me."
Nevertheless, internal Pfizer records recently released by the FDA reveal that is not the case. Indeed, after taking a closer look at documents released by the FDA in March as part of the ongoing FOIA request for background into the agency's approval process for the company's experimental mRNA gene-therapy jab yesterday, Del Bigtree's ICAN legal team reported on serious conflicts of interest.
Nov 25, 2019: Dr. ANTHONY FAUCI said that he wanted the chance to respond to a brand new pandemic.
A 61-page document entitled "Investigational BNT162 Vaccine Program" references Pfizer's equivalent to the FDA's DSMB, a group the pharmaceutical giant dubs its 'External Data Monitoring Committee" (EDMC). Of the five original members listed on page 51 of the document, ICAN notes that two of them are Maternal-Fetal Medicine doctors. While interesting, even more noteworthy is the disclosure that original Pfizer vaccine safety committee member Kathryn Edwards, MD—a professor at Vanderbilt University—was a paid advisor to Pfizer directly before joining the DSMB. ICAN writes:
"[Edwards] has also received payments, speaking fees, or funding from numerous pharmaceutical companies, including Moderna, Merck, and Smith-Kline Beecham, while Sanofi provided her with trips to Paris, Dublin, Amsterdam, and Cancun, among other destinations. Nice work if you can get it!"
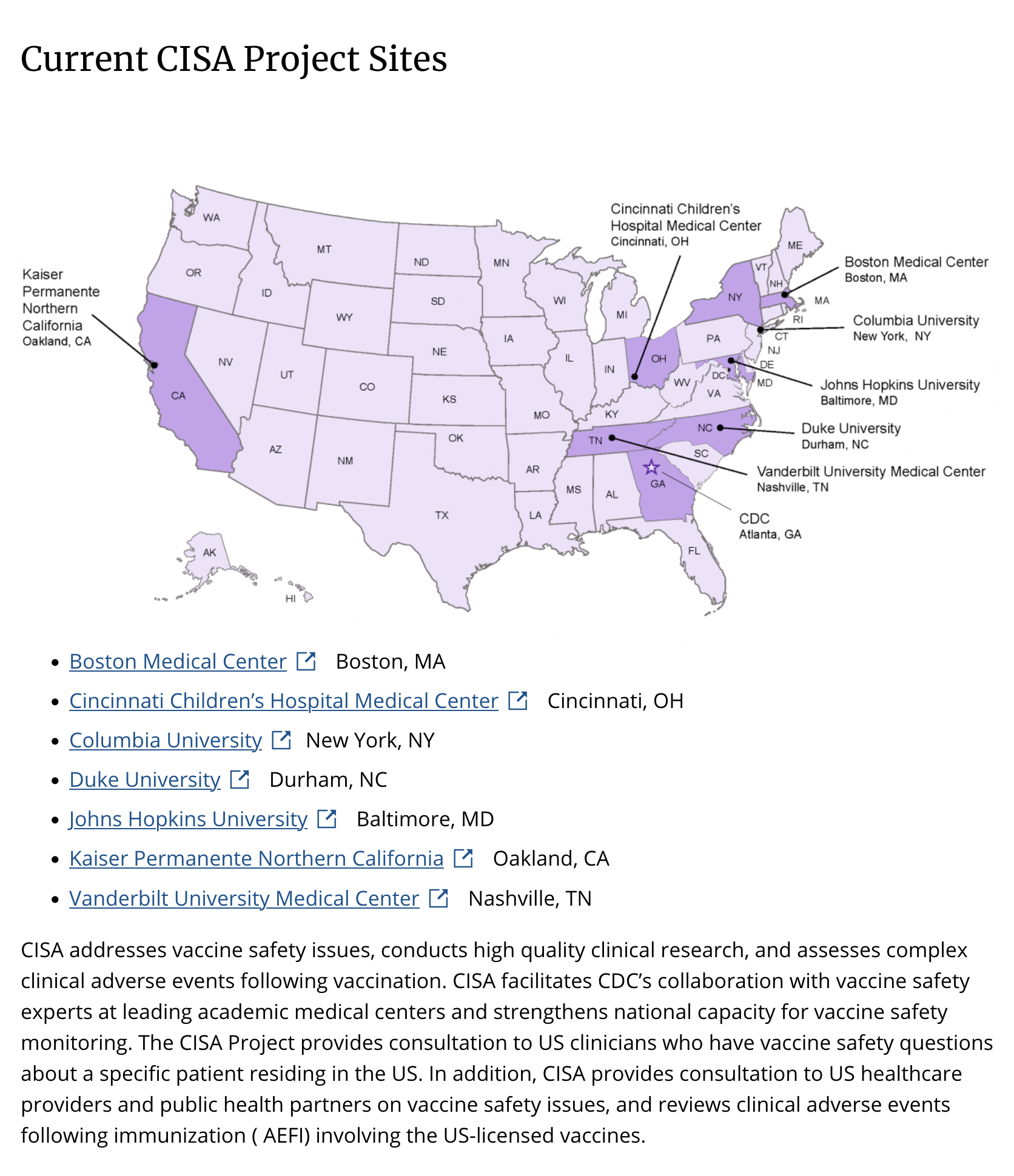
Additionally, the Vanderbilt Hustler reported on Sept 19, 2020, that Edwards leads safety initiatives for vaccines at VUMC along with the Clinical Immunization Safety Assessment Project (CISA) conducted by the CDC. She also sits on the agency's advisory committee (ACIP) on immunization practices. Notably, the CDC's CISA project, in collaboration with Edwards and VUMC (and six other facilities), addresses vaccine safety issues, conducts high-quality clinical research, and assesses complex clinical adverse events following vaccination.
 Screenshot / CDC
Screenshot / CDC
Despite the long list of Edwards' professional activities related to vaccines, ICAN points out that in her July 2020 presentation to the ACIP (titled COVID-19 Vaccine Safety Considerations), she failed to disclose that she had been on a COVID-19 DSMB for Pfizer for over three months at that point.
Of Important significance, Edwards is involved in deciding if Pfizer's experimental shot should be administered to young children. In February—following the discovery that Pfizer's gene-therapy product is less effective in 5 to 11-year-olds—Edwards informed The New York Times that she still recommends the Pfizer "vaccine" for children given the protection against severe disease shown even in the new data set. She explained, "We need to make sure we emphasize the doughnut and not the hole."
Highlighting Edwards' support for vaccinating children against SARS-CoV-2, a Jan. 2022 article in The American Academy of Pediatrics, titled "Proposals to Accelerate Novel Vaccine Development for Children," written by Edwards and colleagues, highlights extensive conflicts of interest in many of the experts, including Edwards, driving the push for "vaccination" as the primary way of approaching COVID-19. Undoubtedly, serious conflicts in this endeavor include prior research funding from the Bill and Melinda Gates Foundation, Merck, Moderna, Pfizer, Roche, Janssen, IBM, Bionet, the NIH, CDC, and several other vaccine-related entities. Still, the authors assert:
"Although most children continue to fare well after SARS-CoV-2 infection, children with underlying comorbidities carry greater risk for severe disease. It was unfortunate that children returned to in-person school in summer 2021 with the youngest lacking access to the most powerful tool we have to protect them against SARS-CoV-2: vaccines."
 Screenshot / American Academy of Pediatrics / Proposals to Accelerate Novel Vaccine Development for Children / Potential Conflict of Interest
Screenshot / American Academy of Pediatrics / Proposals to Accelerate Novel Vaccine Development for Children / Potential Conflict of Interest
Besides highlighting Edwards and her involvement with big pharma and vaccine promotion, ICAN's researchers offered a "thumbnail sketch" of the six other Pfizer's DSMB committee members, touching on the extensive conflicts of interest that exist with them. ICAN reports:
Jonathan Zenilman, MD, of Johns Hopkins University, was the chair of the committee. He received consulting fees from Pfizer in every year from 2014 to 2020 to the tune of $78,279 ($28,168 in 2020 alone). Beyond that, he received $6,045.09 in compensation from being wined and dined, and travelling at Pfizer's expense between 2014 and 2019. His other Big Pharma patrons over these years include GSK, Merck, Smith & Nephew, Cubist, Siemens Medical Solutions, Cipla, Tetraphase Pharma, The Medicines Company, K2M, Becton, Dickinson and Company, Perrigo, and Theravance. Between 2014 and 2020, Dr. Zenilman received an astonishing $91,257.69 from these pharmaceutical companies (including Pfizer) excluding research funding. In addition, Zenilman has received NIH funding.
Robert Belshe, MD, founder of the Center for Vaccine Development at Saint Louis University School of Medicine, received various consulting fees and compensation from Pfizer, GSK, Dermira, Sanofi, AstraZeneca, Merck, Emergent Biosolutions, Comsort, Seqirus, Novartis, and Viiv Healthcare during every year from 2014 to 2020, and from AstraZeneca every year from 2014 to 2018. "How much" you ask? An incredible total of $452,794.26, excluding research funding. Pfizer was especially forthcoming with consulting fees, speaking fees, and generous helpings of "Food and Beverage" and "Travel and Lodging," compensating him $36,225 in 2020 alone. Dr. Belshe disclosed his own conflicts in a 2019 article: Belshe has been a consultant and/or speaker for GSK, Medimmune, Sanofi, Flugen, Novavax, Merck, and Moderna; owns stock/stock options in Flugen; is a scientific board member for Flugen; and has served on data safety monitoring board for GSK, MedImmune, Vical, and Vaxart.
Lawrence Stanberry, MD, of Columbia University, received more than $23,000 from Pfizer in 2020, as well as research funding from the NIH, CDC, and the Bill and Melinda Gates Foundation. He reports consulting fees from Janssen, GSK, and Novartis and is a member of the scientific advisory board of Abivax. Nothing to see here!
Robert Philips Heine, MD, of Wake Forest Baptist Health in Winston-Salem, NC, received $27,450.28 in payments from pharmaceutical companies between 2014 and 2020 for travel and lodging, food and beverage, and consulting fees. Pfizer provided the bulk of these payments with $4,812.50 for consulting fees in 2020 alone. Heine has been a consultant and speaker for GSK and Merck and has received research funding from GSK. His work has been funded by the Bill and Melinda Gates Foundation.
Heather S. Lipkind, MS, MS, of Yale School of Medicine, accepted consulting fees and research funding from Pfizer during 2019 and 2020, for a total of $21,660.
Steve Self, PhD, a statistician at the Fred Hutchinson Cancer Research Center, is also a board member of Health Stream Analytics, an organization developing "novel easy-to-use biometric sensors."
 As the FDA continues to release Pfizer data, UncoverDC has reported numerous times on the potential conflicts of interest surrounding the experimental "vaccines" being promoted under emergency use authorization during the COVID-19 pandemic. Highlighting the secret meetings and unpublished identities of those being paid by drug giants to make decisions about an experimental gene therapy product, ICAN summarized the sentiment held by many watching it unfold, saying:
As the FDA continues to release Pfizer data, UncoverDC has reported numerous times on the potential conflicts of interest surrounding the experimental "vaccines" being promoted under emergency use authorization during the COVID-19 pandemic. Highlighting the secret meetings and unpublished identities of those being paid by drug giants to make decisions about an experimental gene therapy product, ICAN summarized the sentiment held by many watching it unfold, saying:
"We've said it before, but now we're saying it again, and louder: the individuals involved with overseeing and green-lighting COVID-19 injections are a cozy club of insiders whose pockets are now further richly lined with Big Pharma spoils."
Dr. Robert Malone: It's Up to the People to Hold the Perpetrators Accountable


